You may already know proteins as the molecules that can help build muscle. That’s true, but proteins do far more than that. They act as enzymes to speed up reactions, defend the body as part of the immune system, and regulate nearly every process that keeps cells alive.
Proteins also play a big role in cancer. When proteins are misfolded or mutated, they can help tumors grow faster, avoid cell death, and even escape the immune system.
In this post, we’ll explore what proteins are, what each layer of their structure looks like, a few examples of how they operate in cancer cells, and what happens when their environment goes wrong.
From Amino Acids to Complex Structures: What Makes Up Proteins?
Proteins are polymers, large molecules made from repeating units called amino acids. These repeating units are called monomers. An example of a monomer previously covered was nucleotides in DNA. The amino acids are connected in long chains by peptide bonds.
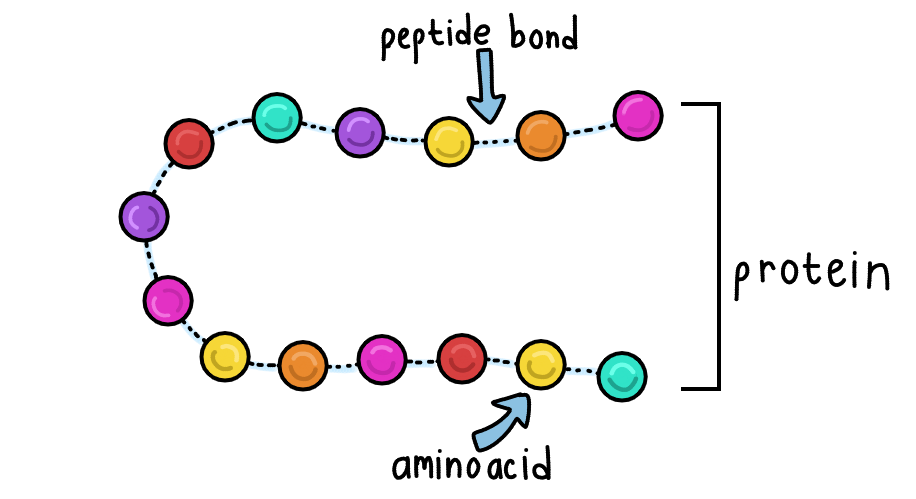
We get proteins from food, break them down into amino acids, and then rebuild them into the specific proteins our cells need. This rebuilding process, called protein synthesis, happens at the ribosomes in the cells.
Each amino acid is made of:
- A central carbon
- An amino group
- A carboxyl group
- A hydrogen atom
- A variable R group
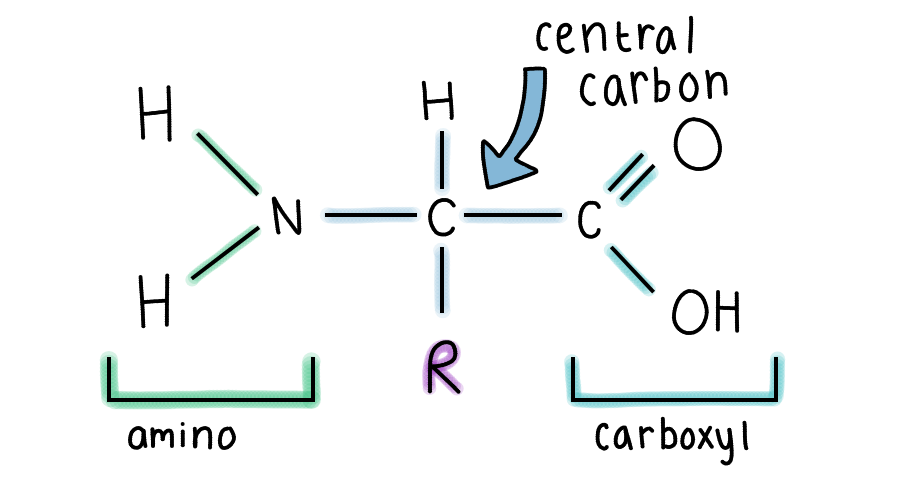
The R group is what makes each type of amino acid unique. Different R groups have different characteristics, like being acidic or non-polar. There are 20 different types of amino acids, and their specific sequence in a polypeptide chain determines how a protein folds and functions.
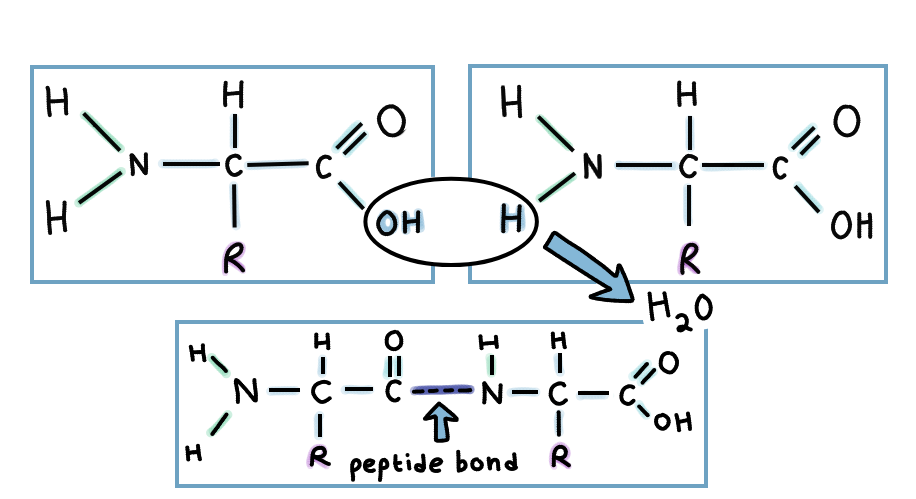
Two amino acids are connected via a peptide bond. This is a type of dehydration synthesis process, called that way because a water molecule is removed from the two amino acids.
Proteins are coded by sections of the DNA called genes. For a refresher on genes, codons, and mutations, here is a previous blog post explaining all of them.
The 4 Levels of Protein Structure
A protein’s function depends on its shape. There are 4 main levels of structures that make up that shape :
- Primary Structure
- Secondary Structure
- Tertiary Structure
- Quaternary Structure
- Primary Structure
This is the exact sequence of amino acids in a chain. The specific order matters because different amino acids interact and form bonds with each other depending on who is near them. The types and prevalence of certain bonds influences the shape and how well the protein functions.
Mutations to DNA can cause codons to change, and proteins to not be built correctly, causing misfolding. Even a single change in the amino acid sequence can alter how a protein folds.
In cancer, mutations often alter this sequence, leading to misfolded proteins that can no longer regulate cell growth properly.
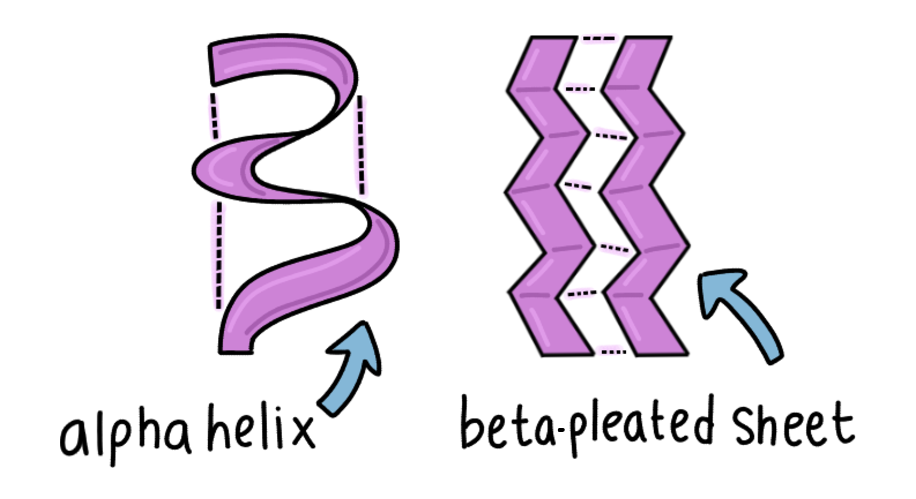
- Secondary Structure
Here, hydrogen bonds form between the carboxyl and amine groups in nearby amino acids, creating repeating structures of :
- Alpha helices
- Beta-pleated sheets
- Tertiary Structure
This is the overall three-dimensional folding of a single polypeptide. In this structure, R groups influence what kinds of bonds occur and how the proteins fold. Some interactions include:
- Van der Waals forces
- Disulfide bridges ( between 2 sulfer atoms of cysteine amino acids )
- Hydrogen bonds
- Ionic bonds
R groups also orient themselves based on their affinity for water. If they are nonpolar/water-hating, called hydrophobic, they are found near the inside of the folded protein. Here, they are less likely to interact with water. If they are polar/water-loving, called hydrophilic, they are found near the outside of the folded protein. This helps stabilize the protein.
- Quaternary Structure
This is where multiple folded protein tertiary subunits come together and connect via binding.
A key example is the p53 protein, often called the “guardian of the genome.” The protein has 4 identical subunits of tertiary structure bound together.
This protein checks the DNA for damage and decides whether the cell should repair itself or self-destruct in a process called apoptosis. In many cancers, p53 is mutated. In many cancers, p53 is mutated, preventing it from functioning and allowing damaged cells to keep dividing.
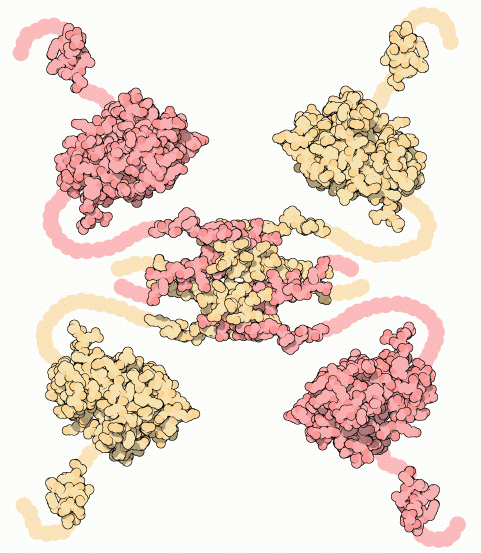
“p53 tumor supressor” by David Goodsell, via RCSB PDB, licensed under CC BY 4.0
Proteins in Action: How They Function in Cancer
Proteins work in all sorts of different places in the cells. Here are 3 especially relevant to cancer:
- Antibodies
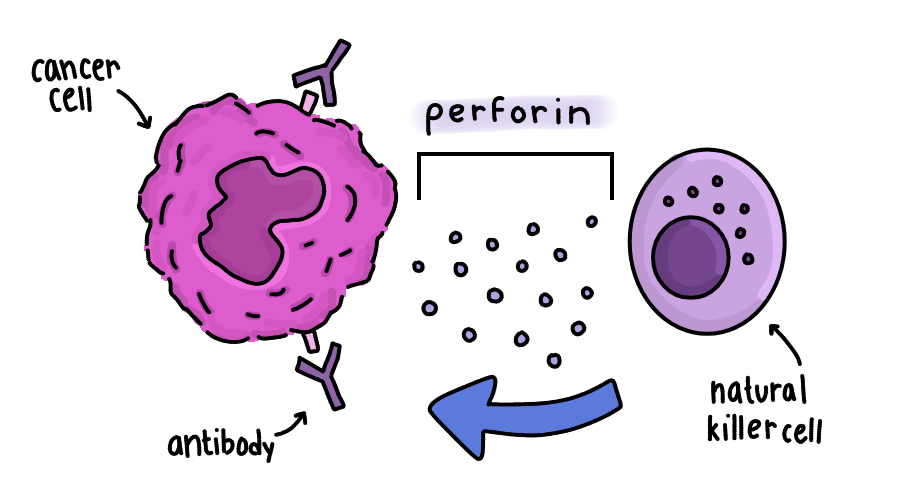
Antibodies are Y-shaped proteins that recognize and bind to foreign or abnormal cells.
In a process called antibody-dependent cell-mediated cytotoxicity (ADCC), antibodies attach to cancer cells, signaling immune cells like natural killer cells to destroy them. These immune cells release proteins like perforin, which cause the cell wall to lyse and the cell to die.
- Enzymes
Enzymes are proteins that help speed up chemical reactions by lowering activation energy. Activation energy is the energy needed to perform a reaction.
They bind to substrates at an active site, forming an enzyme-substrate complex, and can be reused repeatedly.
There are two main types of reactions:
- Anabolic: building larger molecules
- Catabolic: breaking molecules down
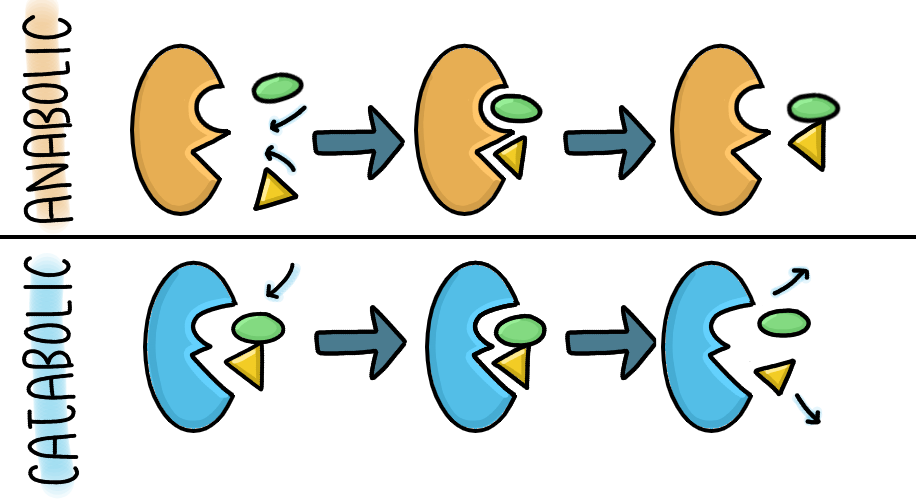
A cancer-related example is hexokinase, an enzyme that converts glucose into glucose-6-phosphate during cellular respiration. Cancer cells often overuse this pathway to meet their high energy demands.
- Protein Pumps
These are proteins in the cell surface membrane. They help transport substances in or out of the cell, sometimes using ATP.
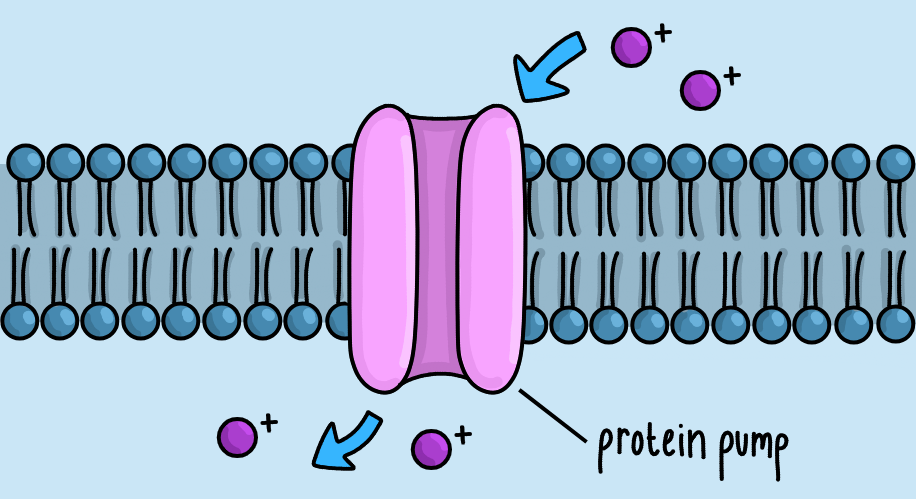
Cancer cells may use protein pumps, like P-glycoprotein, to expel chemotherapy drugs out of the cells using ATP. This limits their effectiveness, contributes to drug resistance, and allows cancer cells to grow back after treatment.
When Conditions Go Wrong: Protein Denaturation
Proteins don’t just work in all conditions. They need the right kinds of conditions to perform their best. If they don’t have these, the proteins can decrease in efficiency or even break down. The process is called denaturation, and it disrupts the bonds that maintain structure, causing proteins to lose function.
Temperature Effects:
- Low temperature reduces kinetic energy ( the energy an object has while it’s moving ), slowing down movement.
- This can also cause something called cold denaturation, where protein bonds are broken, causing the tertiary and quaternary structures to break down.
- Loss of kinetic energy especially affects enzymes because they require enough energy to make the process of colliding with a substrate faster, decreasing the rate of reactions.
- High temperature increases kinetic energy too much, breaking bonds and causing proteins to denature
pH Effects:
- Changes in pH change the charge of the amino acid R groups
- This disrupts bonds and leads to misfolding
Conclusion
Proteins are far more complex than we assume them to be. They are highly specialized and control nearly every aspect of life. From defending the body to fueling cell growth, their functions depend on their structure.
In cancer, this relationship becomes even more important. Small changes in DNA can change protein shape, leading to misfolded proteins that allow cells to grow uncontrollably, resist treatment, or evade the immune system.
In the next post, we’ll explore how these proteins are made in the cell in the process called protein synthesis.
All original insights and illustrations are my own. This content is not intended as medical advice.
Drawings are simplified for illustrative purposes and may not be exact representations of the subjects
Sources
https://medlineplus.gov/genetics/understanding/howgeneswork/protein
https://www.youtube.com/watch?v=HSCUAjZQhXI
https://www.genome.gov/genetics-glossary/Protein
https://www.ncbi.nlm.nih.gov/books/NBK555990
https://www.youtube.com/watch?v=hok2hyED9go
https://www.youtube.com/watch?v=iXX7ZxIbXpA
https://www.youtube.com/watch?v=EweuU2fEgjw
https://www.youtube.com/watch?v=wvTv8TqWC48
https://www.youtube.com/watch?v=O5gN-IK6uKs
https://www.ncbi.nlm.nih.gov/books/NBK22268
https://medlineplus.gov/genetics/gene/tp53
https://pmc.ncbi.nlm.nih.gov/articles/PMC3756401
https://www.verywellhealth.com/the-p53-gene-its-role-in-cancer-2249349
https://byjus.com/chemistry/alpha-helix-and-beta-sheet
https://www.nature.com/articles/s41419-024-06976-0
https://www.sciencedirect.com/science/article/pii/S2772892723000019
https://www.sciencedirect.com/science/article/pii/S2949713225000783
https://www.genome.gov/genetics-glossary/Antibody
https://my.clevelandclinic.org/health/articles/21532-enzymes
https://www.genome.gov/genetics-glossary/Enzyme