Table of Contents:
- Opening
- Who She Is
- How She Got There
- Science She Works On
- How Tumors Outsmart the Immune System
- A Typical Day
- How Scientific Research Actually Works
- Reflection & Advice
- A Paper that Caught Her Attention
- Conclusion
- Sources
Opening
Cancer is usually defined as a disease of uncontrollable growth, caused by mutated cells that turn against the body. What I learned was that it is just as good at hiding from the body and using immune system mechanisms against it to survive.
Megan, a second-year PhD student in molecular cancer biology, is trying to understand a specific subset of this problem. How can immunotherapy, a treatment designed to stimulate the immune system, still work in glioblastoma when the brain has built-in guardrails that suppress immune attack?
In this conversation, we talked about her path from aspiring pediatric oncologist to researcher, how tumors exploit immune system mechanisms, what a cancer biology PhD actually looks like, and how failure, not sucess, might be the most important part of science.
Who She is
Megan is a second-year molecular cancer biology student pursuing her PhD. There, she learns about cancer and pharmacology and combines these 2 interests to conduct her own research.
Megan especially loves cancer biology because it represents a puzzle to her. She enjoys figuring out what a normal biological system looks like, where it breaks, and once it does, if we can fix it.
How She Got There
Megan didn’t always know that she wanted to pursue a Ph.D. At one point, she wanted to go to medical school to become a pediatric oncologist.
After graduating from college, she worked as a research technician in a breast cancer research lab. There, she helped develop a more realistic in vitro ( outside of the body ) assay that combined different cell types in a 3d structure to mimic the tumor microenvironment ( the physical and biochemical surroundings of a tumor).
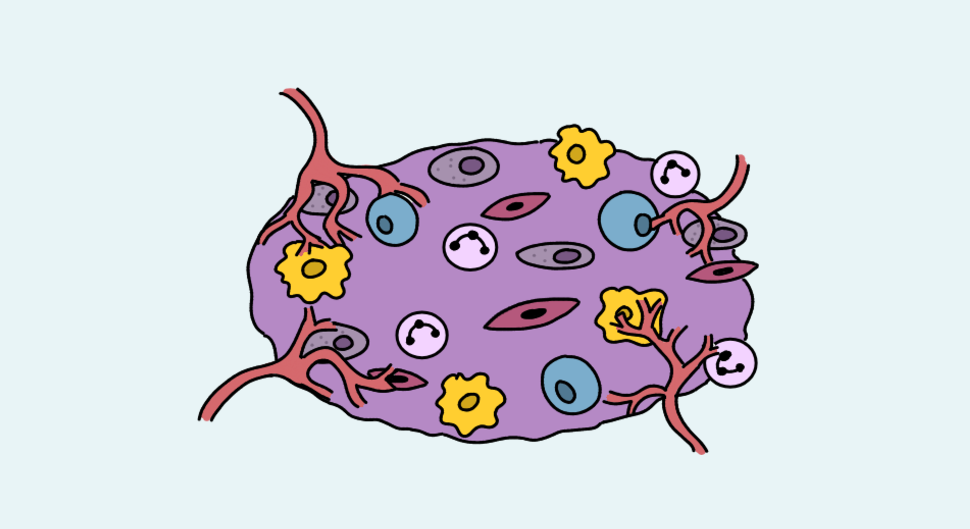
Megan realized how much she loved research because it allowed her to still help patients by generating knowledge to shape future treatments rather than directly treating them. This inspired her to take a chance and apply to the PhD program she is in today.
Science She Works On
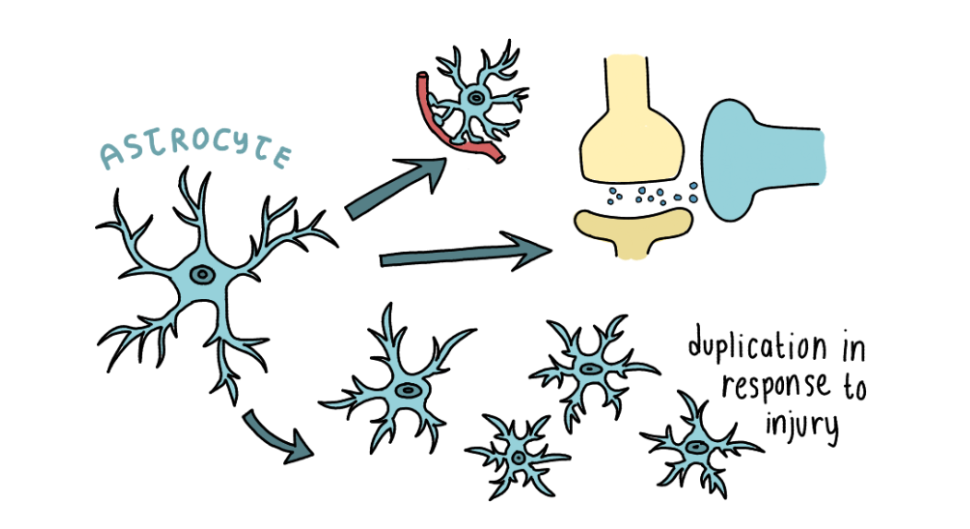
Megan is currently researching a cancer called glioblastoma. This aggressive cancer arises from the brain or the spinal cord, and specifically forms from cells called astrocytes.
These cells conduct tasks such as:
- Controlling blood flow in the blood-brain barrier
- Clearing excess neurotransmitters from synapses
- Responding to injury in the brain
There is currently no clear explanation for how specifically glioblastoma is caused. However, most cancers are caused by changes in the cell’s DNA called mutations. DNA is the code in your cells that tells them how to make proteins to help sustain themselves.
Some examples of this include code for the proteins that help make insulin, hemoglobin, keratin, and more.
DNA tells when cells need to die ( apoptosis ) and when to divide. Mutations, or edits, to this code can cause cancer cells to arise– cells that divide and make other cells quicker, and live when they are supposed to die. The accumulation of these cells forms a tumor.
How Tumors Outsmart the Immune System
Cancer has many mechanisms to ensure survival. Here is a quick overview of some of the main mechanisms that Megan is researching.
One example of a mechanism is hiding. In the immune system, T cells are a type of lymphocyte that help the immune system fight intruders. CD8+ T cells are a type of T cell that destroy cancerous cells. These cells are special because they are focused.
CD8+ T cells recognize small pieces of proteins from inside a cell that are displayed on that cell’s surface. Most healthy cells and tumor cells do this, and T cells use these signals to decide whether a cell is from the body or a foreigner. These proteins are called HLA proteins.
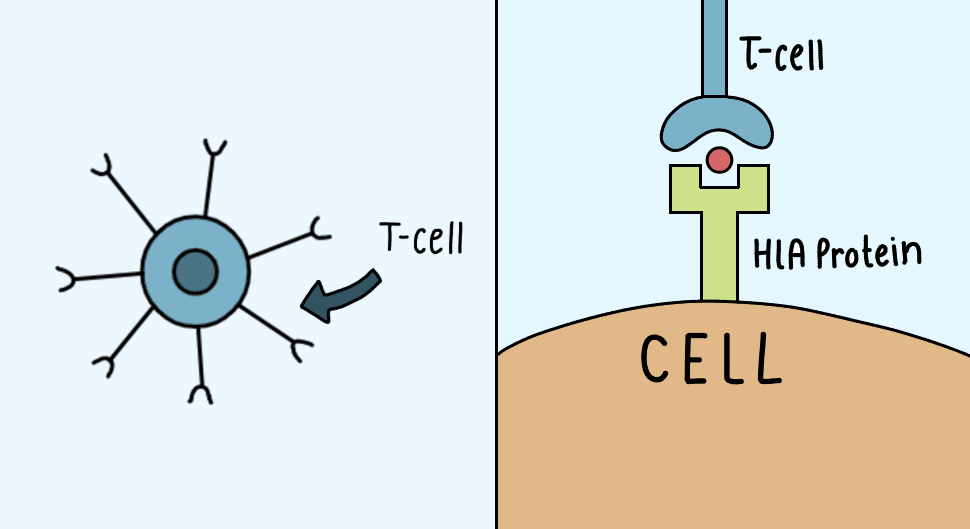
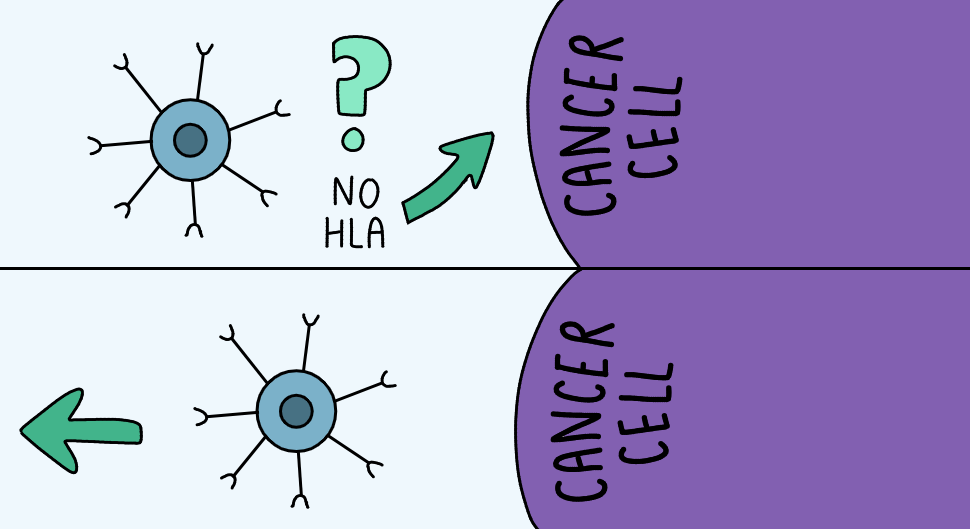
Some cancers reduce HLA expression so that CD8+T cells cannot identify them properly.
Others exploit immune system breaks designed to prevent autoimmunity.
The specific mechanism that most immunotherapy treatments target is the PD-1 pathway. CD8+ T cells have a protein called PD-1 on their surface, which binds to PD-L1 to prevent overactivation of the immune system when fighting a disease.
PD-1 is a protein found on CD8+ T cells, while PD-L1 is often expressed on tumor cells and other cells in the tumor environment.
The tumor microenvironment ( the specific environment surrounding the cancer ) can cause constant stress and have lots of inflammatory signals, causing the CD8+ T cell to be stimulated without a break. This causes exhaustion in the cell and increases PD-1 production.
These tumor infiltrating lymphocytes often resemble chronically exhausted T cells from a long- term unresolved infection. So, when the T-cell finally approaches the tumor cell to kill it, the tumor can express PD-L1 and bind to the PD-1 on the CD8+ T cell, causing a stop signal to be sent to the T cell, rendering it useless.
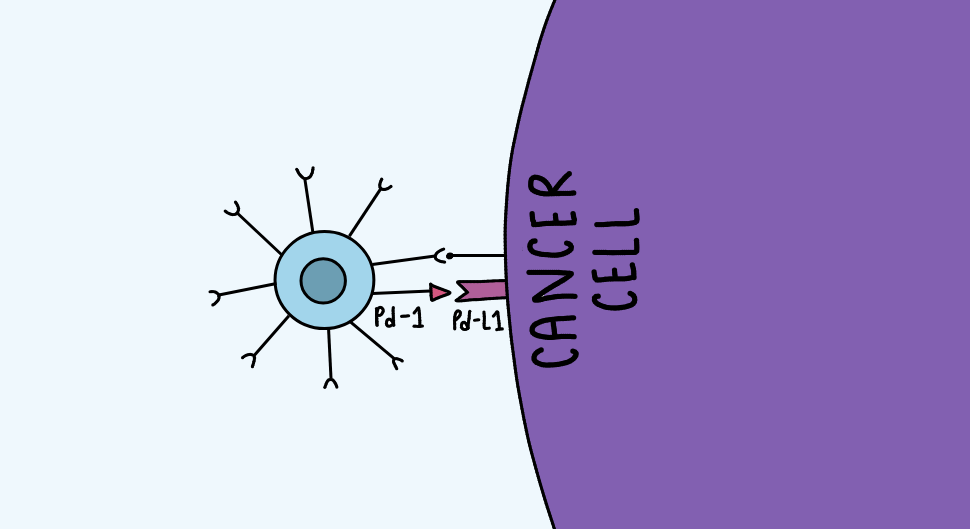
The longer the CD8+ T cell is in the tumor microenvironment, the more PD-1 is made, and the easier it is for the T cell to be suppressed.
An immunotherapy treatment that tries to stop this is anti-PD1 therapies. These try to bind to the PD-1 so that it cannot bind to PD-L1. However, another mechanism prevents it from working successfully in the brain.
This mechanism is called LAG-3. This protein is expressed during prolonged or chronic immune stimulation, similar to what is seen in chronic viral infections like EBV or HIV, where T cells become exhausted over time.
This is especially important in the brain, because it is sensitive, and the immune system must be careful not to cause too much inflammation around neurons. The protein acts as a backup brake for PD-1.
So even if the anti-PD-1 therapy binds to the PD-1 protein, the cell could still be exhausted and not attack the tumor because LAG-3 has been activated.
This is where Megan’s specific research question comes in. How do anti-PD-1 therapies, which don’t work in the brain due to the above reasons, still cause cancer tissue to be killed?
She thinks the answer lies in a current hypothesis about natural killer cells. These are immune cells that kill cells that pose a threat to the body due to an infection or cancer. These are cells that are non-specific, compared to T cells, which are specific. That means they recognize the lack of HLA and attack accordingly.
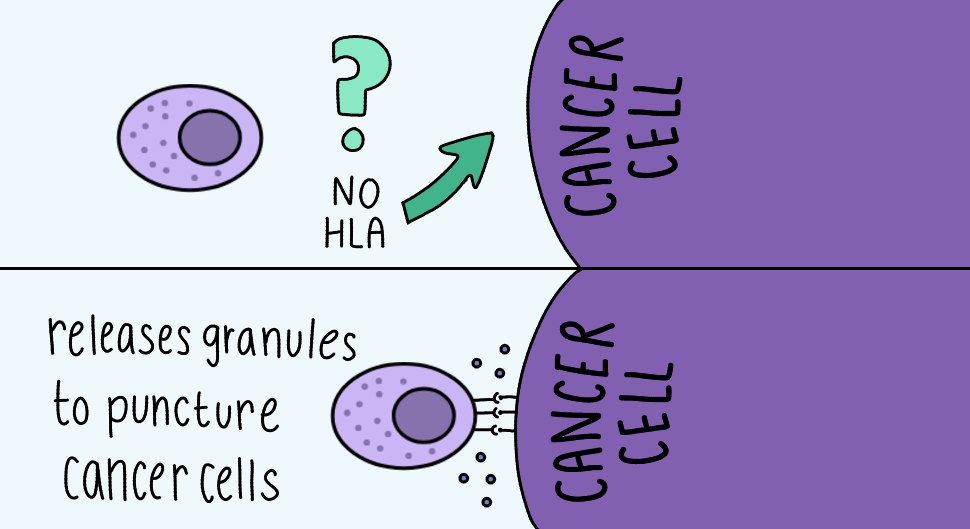
Her research focuses on whether the hypothesis that anti-PD-1 drugs might be activating natural killer cells in ways we don’t fully understand, redirecting them towards tumors, is true.
A Typical Day
Before I talked to Megan, I imagined getting a PhD like conducting your own solo research for 5 years with no additional learning or support. The real experience is much more intricate and interesting.
PhD programs actually work quite differently compared to this. The typical PhD path for cancer biology is often 5-6 years in the United States, and is longer or shorter depending on the country.
However, these years aren’t all about research or experiments. In fact, students have to take classes about their subject matter before even starting their research. They aren’t expected to come into the program with a fully developed question, just the ability to be curious and learn.
Here is a timeline of what to expect when completing a PhD program:
- First-year students study their specific research area and come up with a research proposal by reading past papers published and thinking about what questions need to be answered.
- Second-year students start the data collection and experiment design for their research by working in a lab. They also juggle possibly teaching undergraduate courses and developing their research thesis.
- Third through fifth (or sixth) year students dedicate their time towards researching and conducting experiments about their thesis and completing other tasks such as writing their dissertation or preparing for their thesis defense. This is what you might think of when you envision a PhD student.
Another misconception I had about PhD students is that they were always working around the clock with little time to live a life outside of academia. While students often have to put in extra work to ensure the best research results and prioritize the program, their lives don’t have to revolve around their PhD all the time.
Megan’s usual work week is around 40 hours. She is in her second year and splits her time between attending classes and spending time in the glioblastoma lab researching.
Speaking of classes, these are unlike what classes look like in high school and even at the undergraduate level. PhD classes tend to be more discussion-based rather than lectures, and involve more reading, writing, presenting, and working in groups rather than memorization and exams.
So if you prefer to learn about concepts deeply without worrying whether they will be on a test, a PhD might be a good choice. Also, contrary to popular belief, getting a PhD doesn’t mean solely learning about one subject narrowly.
One thing that surprised me was how interdisciplinary everything is. The program also spans many different disciplines, from molecular biology to computational biology, allowing students to pursue multiple interests.
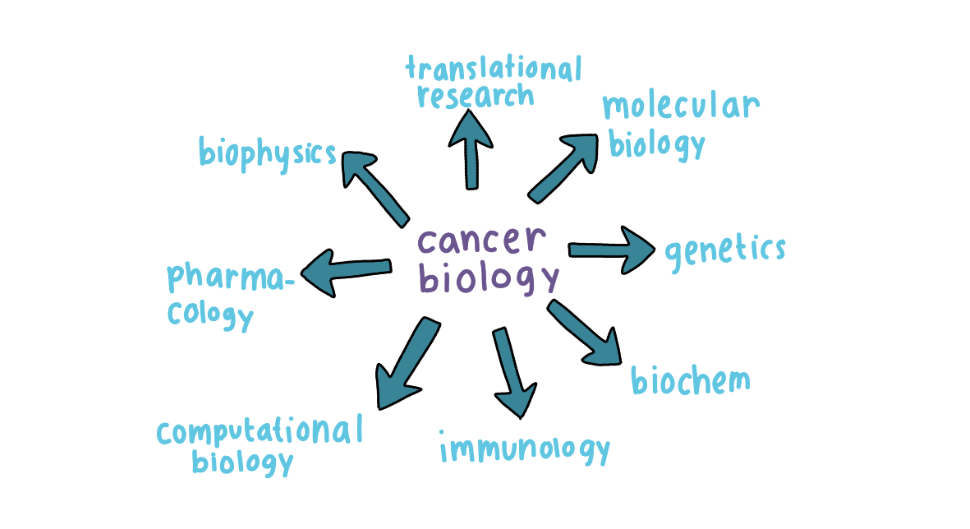
For the research side of the Phd, Megan is currently working on:
- Growing and maintaining specialized cell lines
- Assisting with projects for early-phase clinical trials
- Learning computational techniques like bulk RNA sequencing
- Building the basics of her own research project
When I asked her about what she loved the most about the program, her answer wasn’t about prestige or publications in fancy journals. It was the people.
Being surrounded by other smart and motivated people who are also eager to learn and hopefully change the world too, and listening to seminars where researchers from around the world are some of her favorite parts of getting a PhD. “ You can really nerd out about the tiny details and the huge unanswered questions with others without feeling like you’re bothering me,” she said.
In Megan’s experience, despite all the challenges, she wouldn’t want to be anywhere else because of how deeply rewarding getting her PhD has been.
How Scientific Research Actually Works
Something that Megan really emphasized right from the beginning of the interview was failure in research and learning from those mistakes. Research is messy.
Often in science research, polished projects showcasing significant positive results are published, while projects whose experiments have failed rarely appear. She has seen others, for example, abandon their projects because they didn’t get the data they wanted, instead of learning from their mistakes.
However, she says that research really involves getting comfortable when things fail and deciding to keep on going regardless. In fact, most experiments don’t work on their first try, taking many more attempts and lots of trial and error before something goes right.
It takes grit to pursue research because, oftentimes, it will be very tempting to quit a project.
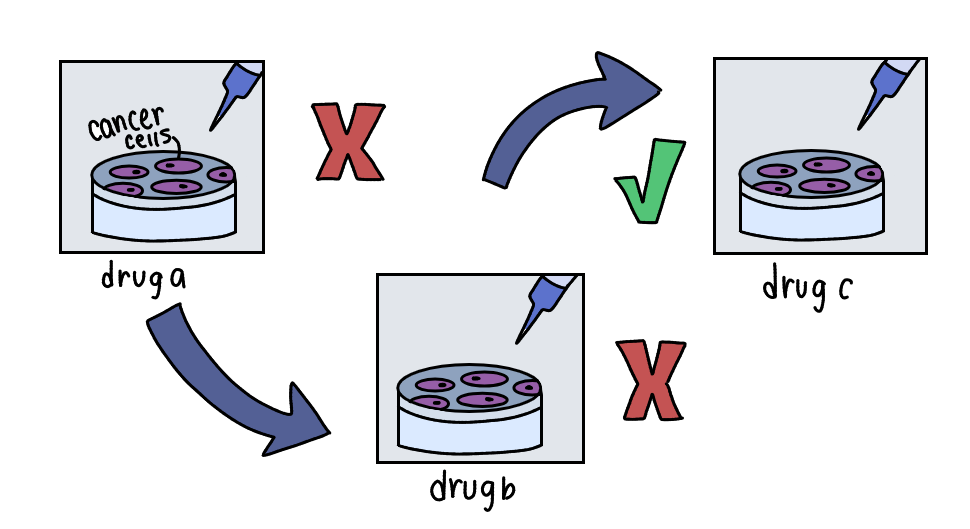
Learning, in Megan’s eyes, involves lots of mistakes and being uncomfortable. Being comfortable with embracing failures and learning from them is very important in this line of work because these often dictate whether a research project works out or not.
Tweaking an experiment, or deciding to approach things from a different angle due to understanding a past mistake, might even mean stronger results at the end of the day. Even if a research proposal flops, though, there is still value in the failure.
Publishing these failures matters just as much as the successes because they allow other scientists to learn from these mistakes– avoiding ideas that have already been proven not to work.
Reflection & Advice
Along with her reflections about failure in research, another piece of advice that she gave about research surprised me. A misconception Megan vocalized was the idea that a Phd is needed in order to work in cancer biology.
When she worked as a research technician, some of her colleagues didn’t hold advanced degrees. Experience, especially hands-on lab experience, can be just as valuable. Positions such as research technicians, or someone who assists in conducting experiments and collecting data, can help advance cancer research without needing extra training.
However, one of the main benefits of a Phd is being able to advance further in academia and unlock more opportunities.
A Paper that Caught Her Attention
A recent paper that caught Megan’s eye asks a clever question. What if we could redirect the body’s existing virus-specific memory T cells to fight cancer?
Memory T cells are a specific kind of T-cell that “remember” past invaders so the body can respond faster the next time it encounters them.
What fascinated me was that these memory T cells are already around different kinds of tumors in both humans and mice, but they aren’t attacking the cancer. So scientists tried a clever solution.
They injected virus proteins into the tumor cells, activating the memory T cells near the tumors. This offered a pro-inflammatory environment that reactivated these cells, causing tumor growth to slow down or even stop in the experimental models.
This technique was especially cool because scientists figured out a way to take something traditionally seen as a threat, viruses, and use the body’s existing natural defenses against them to help fight cancer.
Discoveries like these, the ones that make you go “ Wow, that’s smart,” are for Megan what cancer immunology is really about. Sometimes the best solutions are the ones made by combining previous solutions in an innovative way.
Conclusion
When our conversation ended, what really stayed with me wasn’t how cancer specifically deceivesthe immune system, or the intricacies of glioblastoma. It was really the mindset Megan described.
Cancer research, especially for tricky diseases like glioblastoma, isn’t easy. There will be lots of times when experiments fail, and hypotheses end up useless. And yet the researchers keep on going.
Learning to fail a lot, and learning from those failures is important because it could mean the difference between a project’s success and failure. Sticking with a question a little longer might offer insightful knowledge.
All original insights and illustrations are my own. This content is not intended as medical advice.
Drawings are simplified for illustrative purposes and may not be exact representations of the subjects
Sources
https://my.clevelandclinic.org/health/body/24630-t-cells
https://guide.wisc.edu/graduate/oncology/cancer-biology-phd
https://www.findaphd.com/guides/daily-life-of-phd-student
https://www.mayoclinic.org/diseases-conditions/glioblastoma/symptoms-causes/syc-20569077
https://www.cell.com/current-biology/fulltext/S0960-9822(21)00121-4
https://www.cancer.gov/publications/dictionaries/cancer-terms/def/tumor-microenvironment
https://my.clevelandclinic.org/health/body/24898-natural-killer-cells
https://uk.indeed.com/career-advice/finding-a-job/research-technician